#018: A Tour of All Senolytics Companies.
Unity, Rubedo, Oisin, Senolytic Therapeutics, + more. BioAge / Rubedo / Elevian fund raise.
📡 In this edition of Longevity Marketcap Telemetry
Last Week in Longevity
Longevity Futures
A Tour of All Senolytics Companies
*Disclaimer: None of this information should be taken as financial advice. It is for education purposes only.
Special note:
SENS Research Fundraising: Several prominent donors (Jim Mellon and Michael Antonov included) are matching donations x3 (!) till the end of the year. SENS does incredible basic research work and outreach in longevity / anti-aging. Please consider donating -- your donation will be 3x leveraged! The returns on basic research cannot be overstated.
-Nathan Cheng @realNathanCheng
LongevityMarketcap.com // LongevityList.com // BiohackStack.com
📝 Last Week in Longevity
BioAge raises $90 million Series C. Kristen Fortney’s machine learning drug discovery and aging biomarkers startup raised a $90 million Series C round from a16z, Longevity Fund, Elad Gil, and others. BioAge is aiming to bring their platform-derived BGE-117 and BGE-175 drugs to Phase 2 trials in 1H 2021.
Rubedo Life Sciences raises $12 million seed round. Khosla Ventures, Longevity Fund, and Refactor Capital all participated in the seed round.
Elevian raises $15 million equity financing. Elevian is developing recombinant GDF11, a protein that is expressed in the blood of young animals and has potential regenerative / anti-aging properties. Prime Movers Lab, Bold Capital Partners, For Good Ventures, Kizoo Ventures, Lauder Partners, Longevity Fund, and others participated in the financing.
📅 Longevity Futures
December 19th, 2020: Aubrey de Grey live Q&A at /r/Futurology.
January 26- 28, 2021: 3rd Annual Longevity Therapeutics Conference.Will include speakers from Unity Biotechnology (Ned David), SENS Research Foundation (Aubrey de Grey), Insilico Medicine (Alex Zhavoronkov), Alkahest, Stealth BioTherapeutics, Lineage Cell Therapeutics, Rubedo Life Sciences, Rejuversen, Laura Deming and others. Online. Cost: $1099 - $1899.
Looking for jobs in the longevity industry? Check out longevitylist.com
A Tour of All Senolytics Companies
What are senescent cells?
Senescent cells are cells that lose the ability to divide. This loss of proliferation is caused by damage to the cell including:
Chronic damage (aging): DNA damage, oxidative stress, epigenetic stress, telomere erosion, etc. Notably, somatic cells can only divide a finite number of times before their telomeres shorten to a critical length. (see Hayflick limit, Exceptions: Stem cells, cancer cells, cells that express telomerase, etc.)
Acute damage: Physical injury and wounds, oncogene activation, toxicity (often from cancer drugs), etc.
Senescent cells: good and bad.
Senescent cells are good for you. It is believed that senescence is an evolved cellular mechanism that prevents damaged cells from proliferating as cancer. Senescent cells also play an essential role in wound healing and in early fetal development.
Senescent cells are bad for you. Damaged cells normally undergo apoptosis (suicide) or are cleared by the immune system. But some senescent cells can avoid clearance and accumulate over time. This is harmful for surrounding tissue because senescent cells are still metabolically active and have abnormal gene expression (due to damage). Senescent cells secrete a variety of factors known as senescence-associated secretory phenotype (SASP), which can cause inflammation, fibrosis, and promote cancer.
Most importantly there is strong evidence that senescent cells are associated with age-related diseases and are a fundamental cause of aging.
Anti-aging therapies targeting senescent cells
A landmark discovery by the Jan van Deursen lab (Mayo Clinic. Baker et al, 2016) demonstrated that the removal of senescent cells via a transgene could reverse aging phenotypes and extend the median lifespan in naturally-aged mice.
This breakthrough led to an explosion of researchers and companies developing anti-aging therapies that target senescent cells. Their efforts can be divided into two approaches:
Senolytics: therapies that kill senescent cells.
Senomorphics: therapies that modulate senescent cells and their SASP.
Unfortunately, there are no universal biomarkers for senescent cells. Senescent cells are heterogeneous in their gene expression and SASP secretory phenotype.
And to make matters more complicated, the current biomarkers used to target senescent cells can be found in some healthy cells as well. The main challenge in senolytics is to create a therapy that can kill enough bad senescent cells without killing too many normal cells -- essentially the same problem in cancer.
To date, a total of three senolytic drug therapies have been dosed in human trials to treat diseases by senescent cell clearance. There are encouraging signs these drugs might work -- or at least that they can clear senescent cells in humans (see Mayo Clinic Phase 1 Trial for Dasatinib and Quercetin, Hickson et al. 2019).
A review of all senolytics companies
Elie Dolgin recently wrote an excellent article in Nature on the roughly two dozen companies developing senescent cell therapies.
In this article, I will examine these companies in greater detail to determine which ones are most promising. At the moment, only one company is public (Unity Biotechnology), but more companies will inevitably go public as they progress to clinical trials.
As an investor in the longevity biotech industry you should familiarize yourself with these companies — senolytics is one of the hottest subsectors. The fundamental biology behind senescent cells is compelling and I believe it is likely that at least one senolytics company will find some success.
List of Companies:
Unity Biotechnology
Senolytic Therapeutics
Rubedo Life Sciences
Oisin Biotechnologies
Cleara Biotech
FoxBio
Numeric Biotech
Siwa Therapeutics
Atropos Therapeutics
Deciduous Therapeutics
Dorian Therapeutics
Rejuversen
Oncosence
Senisca
+ others not covered in depth (non-therapeutic, no public information)
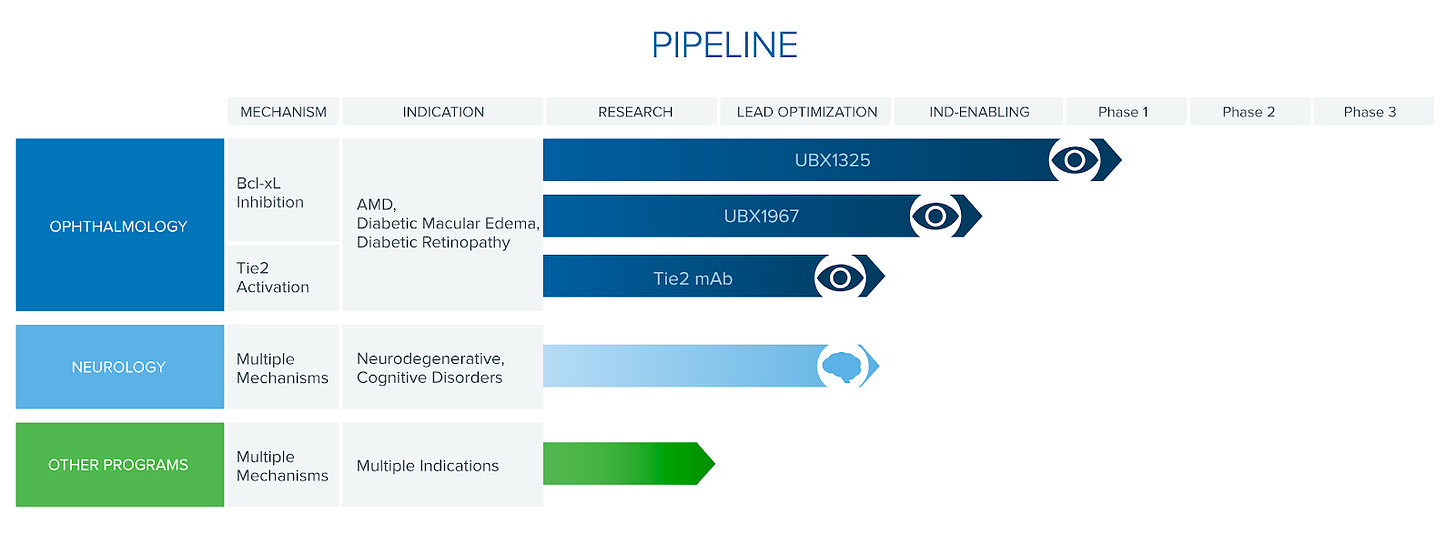
Unity Biotechnology (NASDAQ:UBX)
Market cap: $314 million
Inception date: 2011, IPO 2018
Modality: Small molecule drugs
Pipeline:
UBX1325 // Diabetic Macular Edema // Phase 1
Bcl-xL inhibitor (Bcl-2 family)
Data in 1H 2021
UBX1967 // Eye diseases // Preclinical
Bcl-xL inhibitor (Bcl-2 family)
Fast-follow alternative to UB1325, possible separate development
UBX0101 (Nutlin 3a) // Osteoarthritis (knee) // Phase 2 (failed)
MDM2-p53 inhibitor
Failed August 2020. Possible high placebo effect.
Preclinical Neurology pipeline (TBD)
Lab Lineage: Jan van Deursen (Mayo Clinic), Judith Campisi (The Buck Institute)
Founders: Nathaniel David, Jan van Deursen, Judith Campisi, Daohong Zhou
Notable investors: Longevity Fund, Bezos Expeditions, Founders Fund, ARCH Venture Partners
Links:
General Notes
The flag-bearer of the industry: Unity Biotechnology was the first senolytics company and also the only senolytics company to go public to date. They received $200 million in venture funding and raised $85 million at IPO.
Unity Biotechnology’s current strategy repurposes small molecule cancer drugs to clear senescent cells through a number of different apoptotic pathways (MDM2, Bcl-xL).
Unity’s first drug, UBX0101 failed in a Phase 2 trial to treat osteoarthritis in August 2020. The stock plunged by 66% afterwards.
The company is now focused on its ophthalmology program with a Phase 1 trial for Diabetic Macular Edema (UBX1325) initiated in October 2020.
Origins & Funding
The company was born out of discoveries from the van Deursen lab at the Mayo Clinic and the Campisi lab at the Buck Institute that demonstrated that the clearance of senescent cells could increase the lifespans of mice and reverse aging phenotypes (Baker et al, 2016, Chang et al. 2016).
Nathaniel David, Co-founder: Serial biotech entrepreneur, previously co-founded Kythera Biopharmaceuticals, Syrrx, Achaogen, and Sapphire Energy. Found success by repurposing cancer drugs to cause cell death in fat tissue for cosmetics (Kythera).
Judith Campis, Co-founder: Professor of Biogerontology at the Buck Institute. Made many of the foundational discoveries in senescent cells (biomarkers, SASP).
Jan van Deursen, Co-founder: Former professor at the Mayo Clinic. Demonstrated reversal of aging phenotype with senescent cell clearance in mice. Some drama: Fired from Mayo Clinic for bullying.
Daohong Zhou, Co-founder: Professor of Pharmacodynamics and Radiation Oncology, University of Florida.
Raised $210 million in venture funding from luminaries like Jeff Bezos, Peter Thiel, ARCH Venture Partners, among others.
Raised $85 million in IPO (2018).
Pipeline Details
UBX 1325 Phase 1 trial for Diabetic Macular Edema (DME), an ophthalmologic disease characterized by swollen retinal blood vessels and macula. There is evidence that senescent cells accumulate in the retina and secrete SASP that damages these vessels (Oubaha et al, 2016, Binet et al, 2020).
Preclinical mouse studies showed UBX1325 selectively inhibits Bcl-xL (an anti-apoptotic protein), removes senescent cells in the retina, and improves retinal vasculature. (Unpublished? See slide deck)
Current total market size for DME treatment is ~ $3.7 billion, according to one estimate.
Current standard of care for DME is anti-VEGF therapies that inhibit the growth of blood vessels. Anti-VEGF therapy requires regular injections and they have a slow response (several months to a year). Senolytics can clear cells quickly in a single dose — potentially a superior therapy for DME.
UBX1325, like UBX0101, is also a cancer drug. But UBX1325 targets a different senescent cell pathway: Bcl-xL. They licensed UBX1325 from Ascentage who will receive royalties from commercialization.
UBX1325 preliminary safety and tolerability data expected in 1H 2021. Once completed, Unity will initiate a small proof-of-concept efficacy study in early 2021/2022 with readout in mid-2022. This trial will use objective measurements of disease progression (OCT angiogram), which should be much less prone to placebo effects unlike their osteoarthritis trial.
If UBX1325 runs into issues or seems suboptimal (re: residence time in tissue, tissue exposure), then Unity will switch to their fast-follow alternative UBX1967 (also a Bcl-xL inhibitor) for their ophthalmology pipeline.
More info on Bcl-xL as a target for longevity (Borrás et al. 2020)
Unity also has plans to potentially expand their ophthalmology pipeline indications to include age-related macular degeneration, and diabetic retinopathy.
Outlook for Unity Biotechnology
Unity also has an early pipeline for neurology. Darren Baker’s lab at Mayo Clinic has linked senescent glial cells to tau pathologies in the brain and neurodegenerative disease (Bussian et al. 2018).
Current CEO Anrivan Ghosh was a senior VP and head of research at Biogen, a company known for its efforts in developing Alzheimer’s therapies.
At the Cantor Fitzgerald Healthcare summit in September 2020, CEO Anrivan Ghosh intimated that the company is also exploring different senolytic modalities. Unity has patents for use of senolytic peptides, conjugate-prodrugs, also gene therapy, but these appear to be quite general filings.
Risks
Toxicity as a limiting factor for delivery and dose. Unity’s current approach relies on using toxic repurposed cancer drugs to kill senescent cells. They are basically forced to conduct local injections at low dosage. Industry insiders believe local injections might not be able to counteract the systemic SASP factors produced in senescent cells in the rest of the body. Bcl-xL inhibitors (like UBX1325) are known to kill platelets and there is a risk of thrombocytopenia at high doses.
Cash. According to their most recent quarterly filings, Unity has enough cash to last until mid-2022 ($132 million). This might be just enough to get the readout of their proof of concept efficacy DME trial. The UBX1325 data needs to be convincing enough for investors or lenders.
2nd generation senolytic drugs may prove more effective than 1st gen. 1st gen senolytics were hypothesis-driven repurposed cancer drugs that were known to target certain anti-apoptosis pathways (Dasatinib, Navitoclax, UBX0101, etc). They often have off-target effects and kill some non-senescent cells. Newer 2nd Gen senolytic approaches use high-throughput screens to find drugs that target senescent cells with higher selectivity. 2nd gen senolytics also include targeted strategies like gene therapy, nanoparticle delivery systems, immunotherapy, and prodrugs (galacto-conjugates, etc).
Reward
Diabetic Macular Edema has a large market with a poor current standard of care treatment (anti-VEGFs, requires regular injections).
Possible label expansion for other eye diseases. (DR, AMD)
Unity Biotechnology is the leader in the senolytics industry in terms of clinical development. No other company even has a firm date to enter the clinic while Unity is already trialling their 2nd drug.
Senolytic Therapeutics
Funding: Seed, daughter company of Life Biosciences
Inception: 2017
Modality: Small molecule drug, nanoparticle delivery, monoclonal antibody (immunotherapy), prodrugs
Pipeline:
STX519 // kidney or lung fibrosis? // Preclinical, mature program not publicly revealed
STX434 // kidney or lung fibrosis? // Preclinical, mature program not publicly revealed
STX255 // cancer // Preclinical
Monoclonal antibody targeting senescent cell surface protein, recruits the immune system to clear senescent cells.
Remove chemotherapy-induced senescent cells that cause cancer relapse.
Licensed out to a Swiss investment group. (Rejurveron → Rejuversen).
NephroSEN Project // renal fibrosis (orphan disease) // Phase 2 ready
Phase 2 ready candidate for new target.
Proof of concept development with consortium in Sweden.
Galacto-conjugate pro drugs
Cytotoxic prodrug that is activated in the presence of SA-beta-galactosidase enzyme.
Nanoparticle delivery system
Nanoparticle with holes plugged by beta-galactose. Releases payload in the presence of high levels of SA-beta galactosidase -- biomarker of senescence.
Lab Lineage: Manuel Serrano (IRB)
Founders: Marc Ramis Castelltort, Manuel Serrano
Notable investors: Life Biosciences
Links:
General Notes
Senolytic Therapeutics develops small molecule drug senolytics, senomorphics (SASP blockers), and also immunotherapeutic senolytic approaches.
Also develops prodrug senolytics and nanoparticle delivery systems for more targeted approach.
Developed a senolytics discovery platform using CRISPR.
One of the daughter companies of David Sinclair’s Life Biosciences -- shared expertise, resources, and information.
Based in Barcelona and Boston.
Origins & Funding
Marc Ramis Castelltort, Co-founder and CEO: Ph.D Biochemistry at Oxford, serial biotech entrepreneur (Nineveh Therapeutics, Rejurvesen AG, Chasing Science).
Manuel Serrano, Co-founder: Researcher at IRB Barcelona and one of the discoverers of the p16 tumour suppressor gene / marker for cellular senescence. Also co-founder of Rejuversen.
Funded by Life Biosciences -- David Sinclair’s mega-longevity company. Financing is probably very solid. Life Biosciences raised a $50 million Series B round at a $500 million valuation in 2018 and is looking to raise $100 million this year.
Currently raising money for IND-enabling studies, according to CSO Tim Cash (Simple Biotech podcast).
Pipeline details
Senolytic Therapeutics has two mature preclinical programs that they are readying for clinical trials: STX519 and STX434. Secretive. Details have not been made public but CSO, Tim Cash, has indicated they are one senomorphic + one senolytic drug that target fibrotic lung disease and fibrotic kidney disease. Both are linked to senescent cells.
STX255 is a monoclonal antibody therapy designed to target chemotherapy-induced senescent cells. It is believed the signalling from these cells can cause cancer relapse.
STX255 has been licensed out to Rejuveron / Rejuversen -- a senolytics startup by the same co-founders as Senolytic Therapeutics.
Developing nanoparticle drug delivery systems that release their contents in the presence of SA-beta-galactosidase, a marker of senescence. This is achieved by placing cytotoxic drugs inside a nanoparticle with holes plugged by galactose.
Also developing prodrugs through galacto-conjugation. Essentially attaching a senolytic drug to an acetylated galactose molecule. Senescence cells have high levels of SA-beta-galactosidase, which cleaves the galactose molecule from the senolytic molecule. The senolytic then kills the cell.
Outlook for Senolytic Therapeutics
Optimizing a drug discovery platform by searching for novel targets. Using CRISPR-based genetic screens and others.
Solid team and financing / network from parent company, Life Biosciences.
Fundraising for IND-enabling studies. Mature senolytic and senomorphic programs for lung and kidney fibrosis. Clinical trials seem imminent in the near future.
Mature preclinical senomorphics likely targeting fibrotic (kidney or lung) diseases getting ready for clinical trials -- but no firm timeline yet.
2nd generation senolytic / senomorphic therapies. Definitely furthest ahead in senomorphic development. Their prodrug and nanoparticle delivery systems are interesting. But Tim Cash seemed to suggest that the nanoparticle delivery system was not their main focus, more of a side asset.
Risk
Hard to say what the risks are when so few details about their target and indications are available. It is not even clear what modalities are being used in their mature programs -- though it is likely small molecule.
Reward
Pursuing both senomorphic and senolytic therapies.
Broad modality approaches (small molecules, monoclonal antibody immunotherapy, prodrugs, nanoparticle delivery systems).
Access to Life Biosciences network, resources, and financing.
One of the more developed senolytic companies. Nearing clinical trials.
Rubedo Life Sciences
Funding: Seed, $12 million
Inception: 2018
Modality: Small molecule drugs
Pipeline:
5FURGal prodrug? // (indication?) // Preclinical
Cytotoxic prodrug that is activated in the presence of SA-beta-galactosidase enzyme.
Lab Lineage: Marco Quarta (Stanford)
Founders: Julian Klein, Marco Quarta, Mark Gallop
Notable investors: Khosla Ventures, Longevity Fund, Refactor Capital, Shanda
General Notes
Details on pipeline assets are not public.
Rubedo Life Sciences is possibly developing small molecule prodrugs that only become active in the presence of SA-beta-galactosidase, a senescent cell marker.
Pipeline includes therapies for idiopathic pulmonary fibrosis (IPF) and chronic obstructive pulmonary disease (COPD).
IND-enabling studies expected in 2021.
Developed ALEMBIC, their drug discovery platform to find novel senolytic candidates.
Founder Marco Quarta is also a founder at Turn Bio.
Origins & Funding
Co-founder / CEO Marco Quarta: Research Director at Center for Tissue Regeneration, Repair and Restoration at Stanford University. Also co-founder of Turn Biotechnologies, a longevity startup that focuses on epigenetic reprogramming.
Announced a $12 million seed round funded by Khosla Ventures, Longevity Fund, Refactor Capital, and Shanda on December 3rd, 2020.
Pipeline Details
Developing an unnamed therapy for idiopathic pulmonary fibrosis (IPF) or chronic obstructive pulmonary disease (COPD). Expected IND-enabling studies in 2021.
IPF is a disease linked to senescent cells (Schafer et al. 2017). The global market for IPF treatment is estimated to be $2 billion USD. Current standard of care therapies only slow the progression of the disease.
Also early development in therapies for cancer, sarcopenia, frailty, and liver and kidney fibrosis.
Possibly developing a targeted prodrug that only becomes cytotoxic in the presence of SA-beta galactosidase -- an enzyme elevated in senescent cells. Possibly their lead candidate.
Published preprint on in vivo mouse studies of their 5FURGal prodrug, a 5-fluorouridine chemotherapeutic drug conjugated with beta-galactose (5-fluorouridine-5′-O-β-D-galactopyranoside).
5FURGal prodrug showed to be non-toxic in young mice at equimolar levels that would normally be toxic for 5FUR (160 mg / kg).
5FURGal reduced senescent cells in vitro and in vivo (mice). Also reduced senescent cells induced from doxorubicin, a chemotherapeutic drug.
Aged mice injected with 5FURGal showed improved grip strength, endurance, balance, and spatial learning memory compared to controls.
Mice treated with chemotherapeutic drugs showed less weight loss when treated with 5FURGal compared to controls.
Geriatric mice treated with 5FURGal improved survival (~40%), skeletal muscle functions, stem cell functions, and regenerative capacity.
Outlook for Rubedo Life Sciences
Minimal details on their development timeline available other than IND-enabling studies for their IPF / COPD trial sometime in 2021.
Recently closed a $12 million seed round by Khosla Ventures, Longevity Fund, among others.
Able to generate more novel candidates through ALEMBIC drug discovery platform? No public details on ALEMBIC.
Risk
Hard to assess without many public details. Prodrug approach might require injections, not ideal but not a dealbreaker either. Senescent cells accumulate slowly.
Reward
One of the more developed senolytics companies. IND-enabling studies scheduled for 2021.
Prodrug approach has potential as a highly targeted approach to clear senescent cells.
Applying senolytics in combination with chemotherapy is a promising way to mitigate chemotoxicity and chemo-induced damage. Other companies are pursuing this too (Cleara Biotech).
Oisin Biotechnologies
Funding: Seed, $5 million
Inception: 2014
Modality: Gene therapy
Pipeline:
SENSOlytic Platform // Chronic Kidney Disease (likely orphan) // preclinical, GLP toxicology studies.
Gene therapy using p16 promoter + i-caspase-9 + Protein-lipid vehicle, dimerizer.
OncoSenX (spin out company) // Cancer // preclinical
Solid tumours
Lab Lineage: John Lewis (University of Alberta), CSO
Founders: Gary Hudson, Matthew Scholz
Notable investors: Kizoo Technology Ventures, Methuselah Fund, SENS Research, Hedgewood
Links:
General Notes
Oisin Biotechnologies is developing a novel flexible gene therapy platform to target senescent cells.
Developing a neutral lipid-protein vehicle to deliver a transientDNA payload containing a cell-suicide gene that only activates if the senescenet cell biomarker p16INK4a is transcriptionally activated in the cell. This is achieved with an engineered DNA plasmid containing a p16 promoter and a caspase-9 gene. (Also requires a separate chemical dimerizer).
Oisin’s gene delivery platform technology is cheap, flexible (can target select cells based on gene expression), and scalable. No need to grow viral vectors in cell cultures. Repeat dosing is possible.
Oisin also has an oncology spinout, OncoSenX using the same gene therapy platform technology. OncoSenX seems further along in development.
Backed by SENS Research Foundation / Methuselah Fund.
Origins & Funding
Founded by computer-security-geek-turned-serial-biotech-entrepreneur, Mathew Scholz (founder of Immusoft), and Gary Hudson, a space industry entrepreneur and longtime supporter of SENS.
$2.1 million funding from SENS Research Foundation, Methuselah Fund, Kizoo Technology Ventures, and a few angel investors.
Both Oisin and OncoSenX are licensingEntos Pharmaceutical’s protein-lipid nanoparticle delivery vehicle. Entos is co-founded by John Lewis, Oisin’s CSO.
OncoSenX (oncology spinout) raised $11.4 million, including seed funding from Jim Mellon. They are now looking to raise a $30 million Series A to fund their Phase 1 clinical trials. (Entos received a $4.5 million grant from the Canadian Institutes of Health)
Pipeline Details
SENSOlytic platform based on targeted gene therapy. Uses a neutral protein-lipid nanoparticle (Entos’s Fusogenix) carrying a non-integrating DNA payload (p16 target promoter + inducible caspase-9 apoptosis gene). A separate chemical inducer of dimerization is used to activate the caspase-9 gene and kill the cell. Flexible system, target is interchangeable.
Preclinical results: p16 + p53 targeted therapy extended survival times in mice by 20%. Increased bone density, too. (These are unpublished results).
Safety and distribution: Demonstrated safety in mice, rats, dogs, and non-human primates up to 20 mg / kg. Wide biodistribution. Repeatable delivery.
Currently undergoing GLP toxicology studies.
Clinical trials expected in 1 - 2 years.
Indication: Kidney disease, possibly orphan disease designation.
OncoSenX targeting solid tumour cancers in the lung or kidney. Preclinical studies showed a single systemic dose reduced tumour size by 90% in mice.
Outlook for Oisin Biotechnologies
Current focus is on GLP toxicology studies in preparation for IND. Clinical trials for (rare) kidney disease will follow. No firm date.
OncoSENx is further along and will probably be first to test the protein-lipid vehicle delivery technology.
CEO Matt Scholz mentioned potential applications for companion animals. This makes a lot of sense. Unlike drugs and peptides, which depend on the structure of expressed proteins, Oisin’s approach is more fundamental and operates on expressed genes. I am actually surprised they aren’t currently developing or licensing the technology for pets seeing as they’ve already done tox studies on dogs. Celevity / Rejuvenate Bio / Animal Biosciences could see a major competitor.
Risk
Totally new delivery system. Needs to be fully proven for toxicology and immunogenicity. Also needs good bio-distribution in the human body.
Systemic gene delivery. Risk of accidental integration into the genome even if designed to be transient. Lots of regulatory scrutiny.
Reward
Flexible, low-cost, scalable gene delivery platform technology. Can be easily modified for other transcriptional targets as the science of senescent changes.
Can redose. Important for repeated senescent cell clearance as they accumulate.
Avoids a lot of the issues of viral vectors for gene delivery. (Redosing, package size limits, immunogenicity, laborious manufacturing)
Cleara Biotech
Funding: Seed, unknown
Inception: 2018
Modality: Peptide
Pipeline:
iProx // age-related disease, maybe sarcopenia // preclinical
FOXO4-p53 inhibitor. Analog of proxofim.
iProx // therapy resistant cancer // preclinical
FOXO4-p53 inhibitor. Analog of proxofim.
Lab Lineage: Peter de Keizer
Founders: Tobias Mandl, Marco DeMaria, Peter de Keizer
Notable investors: Apollo Health Ventures, Kizoo Technology Ventures
General Notes
Cleara Biotech is a preclinical senolytics company based in Utrecht.
Developing senolytic peptide therapies that target aging (age-related diseases) and therapy resistantcancer by modulating the FOXO4-p53 pathway.
Born out of the discovery of the FOXO4-DRI peptide by co-founder Peter de Keizer, a researcher now at UMC Utrecht.
Presently designing more selective versions of FOXO4-DRI peptide to be tested in clinical trial.
Funded by Apollo Health Ventures, a longevity-centric venture capital firm based in Germany.
Origins & Funding
Peter de Keizer, Co-founder and CEO: Formerly an academic researcher at Erasmus Medical Center where he discovered the FOXO-p53 pathway for senescence and the FOXO4-DRI therapeutic peptide. He is currently Associate Professor at UMC Utrecht.
Tobias Madl, Co-founder: Associate professor at Medical University of Graz.
Marco Demaria, co-founder: Associate professor at ERIBA (European Research Institute for the Biology of Aging).
Erasmus Medical Center owns the IP for FOXO4 and is developing it through a spinout called Numeric Biotech. de Keizer believed FOXO4-DRI was not ready for human trials and parted ways with Erasmus to develop an improved version of the peptide at Cleara.
Apollo Health Ventures approached Peter de Keizer to start Cleara Biotech based on the strength of de Keizer’s FOXO4-DRI peptide discoveries.
Raised seed funding, but unknown amount.
Pipeline details
Main program: iProx, a more selective version of FOXO4-DRI (proxofim), a peptide that interferes with the FOXO4-p53 interaction. Planned indications: Cancer / Chemotherapy-toxicity, age-related disease (unspecified).
Not many details on prospective trials. Still preclinical.
Likely to go to clinic forcancer trials first. Aging / age-related diseases later.
Most of the science behind FOXO4-p53 senescence was discovered by Peter de Keizer’s lab (co-founder).
Preclinical evidence (Baar et al. 2017):
FOXO4 expression is elevated in senescent cells compared to normal cells.
Inhibiting FOXO4 from interacting with p53 induces apoptosis in senescent cells.
FOXO4-DRI is a D-retro-inverso peptide of FOXO4’s p53 interaction domain. By using D-amino acids (versus the more naturally occurring L-amino acids) the peptide can more easily resist proteolysis -- but still retain the interaction specificity with p53.
FOXO4-DRI has a 11x higher selectivity towards killing (some) senescent cells vs normal cells in vitro. It has better selectivity than dasatinib and Bcl-xL inhibitors like ABT-263 (Navitoclax) in de Keizer’s study.
FOXO4-DRI can reduce chemotherapy toxicity by removing senescence cells damaged by chemotherapeutics (doxorubicin).
Unpublished evidence suggests FOXO4-DRI may extend the lifespan of mice up to 13%.
Outlook for Cleara Biotech
Cleara is still in early stage development. No firm timeline to get to the clinic. No word on IND-enabling studies.
de Keizer has indicated they will likely test their therapy for cancer (combination with a chemotherapy) before an anti-aging trial.
Some people are already self-experimenting with FOXO4-DRI as an anti-aging therapy.
Risk
Numeric Biotech is currently developing the initial version of the FOXO4-DRI peptide while Cleara is focussing on an improved version. Perhaps the original is good enough.
No firm timeline for clinical trials, yet.
Reward
2nd gen senolytic targeting a fairly new apoptosis target (FOXO4-p53).
Potential high selectivity for senescent cells (~10:1).
Peptides are fairly easy to manufacture. D-retro-inverso strategy is promising from a toxicology / resistance to proteolysis standpoint.
For what it’s worth, Reason @ FightAging! Believes this company is one of the better companies in the senolytics space.
FoxBio
Funding: Seed, $10 million
Inception: 2018
Modality: Small molecule drug
Pipeline:
JUV001 // Osteoarthritis // Preclinical (Phase 1 expected 2021)
FOXO4-p53
JUV002 // Aging // Preclinical (Phase 1 expected 2022)
FOX04-p53
Lab Lineage: Kelsey Moody
Founders: Kelsey Moody
Notable investors: Juvenescence, Kizoo Technology Ventures
General Notes
FoxBio is a senolytics joint venture company between Juvenescence and Antoxerene (a daughter company of Kelsey Moody’s Ichor Therapeutics).
Developing small molecule senolytics targeting the FOXO4-p53 interaction.
Phase 1 trials are expected to begin in 2021 and 2022. Indications are believed to be osteoarthritis and aging.
Origins & Funding
Joint venture between Jim Mellon’s Juvenescence and Antoxerene (an Ichor Therapeutics daughter company).
Ichor Therapeutics is Kelsey Moody’s vertically integrated drug discovery company / SUNY-associated research institution / CRO. Very interesting backstory.
$10 million in seed funding. Jim Mellon has said in the past they plan to partner with other pharmas as well.
Pipeline Details
Very few public details. Appears they find drug candidates via high-throughput screens using a FOXO4-p53 assay they developed.
One of the slides in Jim Mellon’s talk at EARD 2020 showed two FoxBio drugs in development to treat: 1. Osteoarthritis and 2. “Aging”.
Osteoarthritis is a ~$7 billion USD global market. Current treatments include corticosteroid injections and acetaminophen.
Phase 1 clinical trials expected to start in 2021 and 2022.
Senolytic small molecule drugs that target the FOXO4-p53 interaction -- a biomarker of some senescent cells.
FOXO4-p53 senescence pathway was discovered by Peter de Keizer, researcher at UMC Utrecht and founder of Cleara. (Baar et al 2017). Inhibiting FOXO4 in mice “counteracted doxorubicin-induced chemotoxicity and restored fitness, fur density and renal function in models of ageing”.
Outlook for FoxBio
Juvenescence (FoxBio’s parent company) is planning to go public via IPO within 6 months, according to Jim Mellon.
Begin phase 1 clinical trials in 2021 / 2022.
Risk
Competing with two other companies targeting FOXO4-p53 (Numeric Biotech, Cleara Biotech).
Standard drawbacks with small molecule drugs: May have greater chance of off target effects and toxicity compared to some other senolytic approaches (peptides, gene therapy, prodrugs)
Reward
FOXO4-p53 is a relatively new senescence pathway (de Keizer et al. 2017). Therapies developed for this pathway may be more selective for senescent cells than drugs targeting Bcl-xL or MDM2 (Navitoclax, etc).
Juvenescence backing. Access to their network of expertise, financing, etc.
Management is competent and resourceful. Kelsey Moody started out by building his own biotech lab in his apartment. Now his company owns its own research facility and graduate students can earn their SUNY degrees doing lab work at Ichor Therapeutics.
Small molecule drugs are cheap to produce, easy to administer, and regulatory path is well-trodden.
Numeric Biotech
Funding: Seed, 300k €
Inception: 2017
Modality: Peptide
Pipeline:
NBT-103 // neuroinflammation or autoimmune disease// Preclinical, clinical trials Q2 2021 expected
FOXO-p53 peptide, aka FOXO4-DRI
Lab Lineage: Peter de Keizer
Founders: Hans van Berkum
Notable investors: UNIIQ
General Notes
Numeric Biotech is a spinout from Erasmus Medical Center that is developing the original FOXO4-DRI senolytic peptide discovered by Peter de Keizer. Numeric is a competitor to de Keizer’s Cleara Biotech.
Focussing on neuroinflammatory and autoimmune diseases.
Clinical trials planned for Q2 2021.
Origins & Funding
Spinout from Erasmus Medical Center, Rotterdam. Based on Peter de Keizer’s discovery of FOXO4-p53 and the FOXO4-DRI peptide. de Keizer disagreed with Erasmus, believing FOXO4-DRI was not selective enough for the clinic.
300k € funding from UNIIQ (investment consortium of EU and universities in Holland)
Hans van Berkum, CEO and Co-founder: Previously CEO at Virosome and management at AstraZeneca and Sanofi.
Pipeline Details
NBT-103: FOXO4-DRI senolytic peptide (proxofim) that inhibits the FOXO4-p53 interaction.
Target indications: Neuroinflammatory diseases and autoimmune disease. Focus on senescent cell-related pathologies that exhibit an interleukin-6 secretory phenotype. Also a focus on using FOXO4-DRI to upregulate functional regulatory T-cells.
Clinical trials expected in Q2 2021.
Background on FOXO4-DRI (same as Cleara Biotech, Barr et al. 2017):
FOXO4 expression is elevated in senescent cells compared to normal cells.
Inhibiting FOXO4 from interacting with p53 induces apoptosis in senescent cells.
FOXO4-DRI is a D-retro-inverso peptide of FOXO4’s p53 interaction domain. By using D-amino acids (versus the naturally occurring L-amino acids) the peptide can more easily resist proteolysis -- but still retain the interaction specificity with p53.
FOXO4-DRI has a 11x higher selectivity towards killing (some) senescent cells vs normal cells in vitro. It had better selectivity than dasatinib and Bcl-xL inhibitors like ABT-263 (navitoclax) in de Keizer’s study.
FOXO4-DRI can reduce chemotherapy toxicity by removing senescence cells damaged by chemotherapeutics (doxorubicin).
Unpublished evidence suggests FOXO4-DRI can extend the lifespan of mice up to 13%.
Risk
FOXO4-DRI (NBT-103) might not be selective enough against senescent cells. It kills roughly 10 senescent cells for every one normal cell.
Directly competing with Peter de Keizer’s Cleara Biotech, that is developing a more selective version of FOXO4-DRI
Reward
Mature development, lead candidate already selected. Clinical trials slated for Q2 2021.
Peptide approach to inhibiting FOXO4-DRI may be more selective than small molecule drugs.
Siwa Therapeutics
Funding: $1.6 million
Inception: 2006
Modality: monoclonal antibody, immunotherapy
Pipeline:
SIWA 318H // senescent cells, cancer // preclinical
Monoclonal antibody that targets cells that have higher levels of glycolysis (cell surface antigen)
Lab Lineage: ?
Founders: Lewis S. Gruber, Misty Gruber
Notable investors: ?
General Notes
Siwa Therapeutics is developing their 318H monoclonal antibody immunotherapy that targets senescent and cancerous cells through an undisclosed antigen marker of increased glycolysis.
Potential applications for senescent cells, cancer, and viral infection.
No public details on timeline for clinical trials.
Origins & Funding
Lewis Gruber, Co-founder CEO: Formerly CEO of ZelleRX, Arryx, and Hyseq.
IP was developed in house from work Gruber started in graduate school.
Pipeline details
Siwa Therapeutics 318H monoclonal antibody immunotherapy to treat cancer and some to be determined age-related diseases. Also developing 318H to treat virally infected cells (COVID-19).
318H binds to cells that have an antigen marker of elevated glycolysis and recruits the immune system to destroy the cells. Some senescent cells and most cancer cells have elevated glycolysis (“Warburg effect”).
Preclinical studies and results:
66% reduction in p16INK4a+ senescent cells in old mice treated with 318H. Increase muscle mass in a naturally aged mouse model
30% reduction in metastasis in triple-negative breast cancer mouse models. “Trending towards reduced tumour size”. (This kind of language is always suspicious)
There is some evidence in the literature for senescence-associated cell-surface antigens. Frescas et al (2017), Kim et al (2017)
Outlook for Siwa Therapeutics
Completed tolerability studies in non-human primates and preparing IND applications.
Collaboration with an unnamed university to develop 318H for pancreatic cancer.
Likely to advance their cancer pipeline first before age-related disease pipeline.
Risk
Many details have not been made public, so it is difficult to assess. Many questions: What is the surface receptor? Response variability? Immunogenicity etc.
Reward
Immunotherapy is very hot in biotech presently -- and with good reason (many successes in cancer therapy). Immunotherapy might be translated to work for senescent cell therapies -- as long as there are targetable surface antigens for senescence.
Atropos Therapeutics
Funding: $339,657, NIH funded
Inception: 2018
Modality: small molecule drugs
Pipeline:
FATES senescence suppressor / stimulator screening platform
Age-related diseases and cancer.
Lab Lineage: Andrew Koff
Founders: Andrew Koff, William Tap
Notable investors: Incite, LC Global Partners
Links:
General Notes
Atropos Therapeutics is developing small molecule drugs that prevent quiescent cells from becoming senescent. (i.e prevent Senescence After Growth Arrest).
Developed FATES (First ATRX-based Therapeutic to Elucidate Senescence, a cell-based phenotypic screen that can identify compounds that suppress senescence (for age-related disease) or stimulate senescence (for cancer).
Collaboration with Estée Lauder to develop senomodulators.
Origins & Funding
Andrew Koff, Founder: Researcher at Memorial Sloan Kettering Cancer for 25 years. Started the Cell Cycle Research lab there. Subject expert on senescence after cell cycle arrest, CDK4 inhibitors for cancer.
William D. Tap, Co-founder: Chief of the Sarcoma Medical Oncology Service at Memorial Sloan Kettering Cancer Center.
$339,657 grant by the NIH.
Pipeline details
Early development. Presently, performing high-throughput screens to find senomorphic drugs that prevent the transition of cellular quiescence to cellular senescence.
Partnering with AI startup Atomwise to find senomodulators.
Cosmetics focussed pipeline in collaboration with Estée Lauder.
Background science / approach (Kovatcheva et al. 2017):
Use drugs to target the ATRX foci pathway. ATRX is an enzyme that remodels chromatin and is also required for chemotherapy-induced senescence of the cell from a quiescent state (temporarily non-proliferative).
General strategy is to slow down the accumulation of senescent cells rather than complete elimination in order to preserve beneficial aspects of senescent cells (wound healing).
Exploring modulation of senescence -- potential for treatment of cancer via senostimulators.
Outlook for Atropos Therapeutics
Still early development and still finding candidate drugs through their FATES screening platform.
Partnership with AI drug discovery company, Atomwise, to find novel senomodulators.
Collaboration with Estée Lauder to develop senomodulators for cosmetics.
Risk
Target indications: Slowing down aging by slowing senescent cell accumulation means more difficulty in choosing an indication / endpoint for a clinical trial.
Reward
Unique approach of targeting senescent cell formation rather than removal.
Deciduous Therapeutics
Funding: unknown, $5.8 million
Inception: 2018
Modality: Immunotherapy, small molecule drug?
Pipeline:
Immunomodulators to clear senescent cells that cause age-related and non-age-related disease. No public details.
Lab Lineage: Anil Bhushan
Founders: Anil Bhushan, Robin Mansukhani
Notable investors: Longevity Fund, crv, 8VC, Presight Capital
General Notes
Deciduous Therapeutics is developing immunomodulators that enable the immune system to naturally clear senescent cells.
Based on the work of Abil Bhushan (UCSF) on senescent cells and immune surveillance. Likely target indication will be T1 diabetes?
Backed by Longevity Fund, 8VC, among others.
Mostly in stealth mode. No public details on their development currently.
Origins & Funding
Anil Bhushan, Scientific Co-founder: 16 years as professor at UCLA and UCSF studying the role of senescent cells in aging, autominnuminty, and metabolic diseases (especially T1 diabetes).
Robin Mansukhani, CEO Co-founder: Serial entrepreneur. Previously co-founded Alzeca Biosciences (MRI diagnostics for neurodegenerative disease) and Cense Biosciences (insulin delivery technology).
$5.8 million seed funding from Longevity Fund, crv, 8VC, Presight Capital
Pipeline Details
Immune modulators to treat diseases associated with senescent cells. Still in stealth mode. Anil Bhushan’s research work focuses on senescent cells and Type 1 diabetes -- this will likely be a target indication.
I couldn’t find any of Anil Bhushan’s published work on senescent cell clearance via immune system approaches. Most of his potential translational work with senescent cells relate to Type 1 Diabetes, which is an autoimmune disorder.
Scientific background, Senescent cells and Type 1 Diabetes (Thompson et al 2019):
In Type 1 diabetes (T1D) beta cells that make insulin in the pancreas are mistakenly destroyed by the immune system.
Beta cells become senescent in T1D and secrete a phenotype that induces senescence in surrounding cells and causes the immune system to attack them.
Inducing apoptosis in senescent cells beta cells with Bcl-2 (marker of senescence) inhibitors prevented T1D in non-obese diabetic mice models.
Outlook for Deciduous Therapeutics
Still in stealth mode, no timeline on clinical trials, IND-enabling studies, or lead programs.
Risk
Hard to assess given lack of public details. Potentially: Variability of responses in immunotherapies.
Reward
Leverage immune system to naturally clear senescent cells. Potentially minimize collateral damage to beneficial senescent cells or non-senescent cells.
Dorian Therapeutics
Funding: unknown,
Inception: 2018
Modality: small molecule drugs
Pipeline:
T001 Senoblocker // Osteoarthritis // Preclinical
Prevent cellular senescence and promote stem cell renewal via USP16 (deubiquitinating enzyme) inhibitor
Senoblockers for CAR-T Cells // CAR-T cell cultures // Preclinical
Improve CAR-T cell fitness and reduce senescence via USP16 inhibitor
Lab Lineage: Maddalena Adorno, Benedetta di Robilant
Founders: Maddalena Adorno, Benedetta di Robilant
Notable investors: Ycombinator
General Notes
Dorian Therapeutics is developing USP16-inhibitor senoblocker to prevent cellular senescence to treat age-related disease and also improve cell viability in CAR-T cell therapy.
Founded by two researchers at Stanford. Incubated by Ycombinator (S18) and StartX
Very early stage. No timeline for clinical trials, yet.
Origins & Funding
Maddalena Adorno, CEO and Co-founder: Formerly a post-doctoral researcher at Stanford University studying stem cell and aging biology. Co-founded Stemguard.
Benedetta di Robilant, CSO and Co-founder: Formerly a post-doctoral researcher at Stanford University at the Institute for Stem Cell Biology and Regenerative Medicine (ISCBRM).
Ycombinator Alum S18. Also an alum of StartX accelerator.
Granted exclusive patent of T001 senoblocker invented by co-founders while they were at Stanford.
Pipeline Details
Developing T001, a “senoblocker” that inhibits the USP16 deubiquitinating enzyme to prevent or delay cellular senescence and improve stem cell renewal.
Target indication: Osteoarthritis. Also applications for improving cell viability in CAR-T cell therapies.
Ubiquitin is a protein found in all cells. It attaches to other proteins to modify their function, location, or to mark them for degradation. USP16 removes ubiquitin proteins from other proteins, most notably from H2A histones -- proteins involved in DNA expression and repair.
Upregulation of USP16 has been linked to cellular senescence and defects in somatic stem cells via the Wnt pathway. (Adorno et al. 2018)
CAR-T application: Ex-vivo expansion of patient T-cells causes accelerated senescence. In this preprint, the founders demonstrated that modulating USP16 decreased markers of senescence of CAR-T cells in vitro, increased cellular fitness, and produced T-cells 60% more efficient in controlling tumor growth in a mouse model of leukemia and neuroblastoma.
Outlook for Dorian Therapeutics
No timeline for clinical development yet. My guess: CAR-T application likely sooner than osteoarthritis therapy.
Entered into an agreement with Ubiquigent to access their library of novel ubiquitin modulating compounds. Likely doing high-throughput screens.
Risk
Harder to design clinical trials for therapies that slow down senescence versus ones that clear senescent cells.
Reward
Application for senoblockers to improve CAR-T cell viability and fitness.
Dorian’s senoblocker potentially has stem cell renewal benefits.
Rejuversen
Funding: Series A, unknown amount.
Inception: 2020
Modality: Monoclonal antibody
Pipeline:
PD-L2 targeting monoclonal antibody // Cancer (combination therapy) // Clinical trials planned in 2023+
Licensed STX255 from Senolytic Therapeutics
Lab Lineage: Manuel Serrano
Founders: Marc Ramis Castelltort, Manuel Serrano, Tim Cash
Notable investors: Rujeveron
Links:
General Notes
Rejuversen is developing a monoclonal antibody targeting PD-L2 in order to recruit the immune system to clear senescent cells that promote cancer relapse or metastasis.
Co-founders include Marc Ramis Castelltort, Manuel Serrano, Tim Cash -- all team members from Senolytic Therapeutics.
Investment by Rejuveron, a Swiss-based investment company.
Origins & Funding
Based on discoveries in senescent cells and cancer by Manuel Serrano (Institute for Research in Biomedicine) in collaboration with Cnio and VHIO. IP for the antibody drug was originally created at Senolytic Therapeutics (STX255), but licensed to Rejuveron / Rejuversen.
Marc Ramis Castelltort, Co-founder and CEO: Ph.D Biochemistry at Oxford, serial biotech entrepreneur (Senolytic Therapeutics, Nineveh Therapeutics, Chasing Science).
Manuel Serrano, Co-founder: Researcher at IRB Barcelona and one of the discoverers of the p16 tumour suppressor gene / marker for cellular senescence. Also co-founder at Senolytic Therapeutics.
Tim Cash, Co-founder: Also currently CSO at Senolytic Therapeutics.
Series A investment by Rejuveron, an investment company based in Zurich, Switzerland.
Pipeline Details
Monoclonal antibody targeting the PD-L2 surface receptor of chemotherapy-induced senescent cells in tumours in order to recruit the immune system to clear the cells.
Chemotherapy can induce senescence in tumour cells (Ewald et al. 2010). These senescent cells can secrete SASP factors that promote tumour initiation and metastasis. (See Wang et al. 2020, Faget et al. 2019)
Several studies by Manuel Serrano showed that senolytic drugs used in combination with chemotherapeutics (cisplatin, palbociclib) enhanced tumour inhibition in mice. (González‐Gualda et al. 2020, Muñoz‐Espín et al. 2018)
PD-L2 (Programmed Death Ligand 2) is a cell surface receptor that negatively regulates the adaptive immune system. According to Serrano (see patent), PD-L2 is highly expressed in senescent tumour cells and damaged cells.
Licensed Senolytic Therapeutics STX255 monoclonal antibody to inhibit PD-L2, thereby allowing the immune system to detect and remove senescent cells.
Outlook for Rejuversen
Castelltort indicated they expected to initiate clinical trials in 3 years.
The patent for their PD-L2 monoclonal antibody seems to suggest expansion for age-related diseases and even the clearance of skin moles.
Risk
Possibly will face the same challenges associated with immunotherapies in cancer: Variability in response, immunogenicity risk, etc.
Reward
Immunotherapy has proven to be a successful approach to kill cancer cells and it is reasonable to hope it could work for senescent cells too.
Developing a treatment for cancers prone to relapse and metastasis --high unmet need.
Strong team and scientific co-founders.
Oncosence
Funding: Unknown
Inception: 2019
Modality: Small molecule drug
Pipeline:
Senescence stimulator + Senolytic // (Liver?) Cancer // Preclinical
CRISPR and chemical screen to find senostimulators + senolytics.
Lab Lineage: Rene Bernards, Andrea Alimonti
Founders: Rene Bernards, Andrea Alimonti
Notable investors: ?
General Notes
Oncosence is developing a two-step therapy (“one-two punch”) that first induces senescence in cancer cells followed by a senolytic to clear them.
Spinout company from Rene Bernard’s work at the Netherlands Cancer Institute (NKI).
Still in early development, not many public details.
Origins & Funding
Spinout company from Rene Bernard’s work at the Netherlands Cancer Institute (NKI).
Rene Bernards, Co-founder: Professor of Molecular Carcinogenesis at the Netherlands Cancer Institute since 1992.
Andrea Alimonti, Co-founder: Professor of Experimental Oncology at ETH Zurich. Group Leader at the Institute for Oncology Research (IOR)
Rolf Jan Rutten, CEO: Formerly founder and CEO of Audion Therapeutics, founder at thromboDX (acquired by Illumina), and DyblyAG.
Pipeline Details
Developing small molecule drugs that induce senescence in cancer cells followed by a second drug that specifically clears the resulting senescent cells. (“One-two punch”)
Preclinical evidence (Wang et al 2019) - Liver cancer:
XL413 (CDC7 kinase inhibitor) induces senescence. AZD8055 (mTOR inhibitor) kills XL413-induced senescent cells.
Used a kinome genetic screen (CRISPR-based) to find 14 kinase genes that are necessary for proliferation of cancerous liver cells that have a TP53 mutation.
XL413, a CDC7 DNA replication kinase inhibitor, was identified through a screen for inhibitors of the 14 crucial kinase genes. XL413 induces senescences in liver cancer cells with a TP53 mutation.
A chemical screen demonstrated that mTOR inhibitors could act as a specific senolytic of cancer cells seno-induced by XL413.
Experiments in liver cancer mouse models showed the combination therapy of the senostimulator XL413 followed by an mTOR inhibitor (AZD8055) resulted in a significant decrease in tumour volume and increased survival.
Global liver cancer market is expected to grow to $11 billion by 2025, according to some reports. 5-year survival rates for liver cancer is 33%.
High-throughput screen (platform technology) to find novel senescence inducing compounds (Wang et al 2017)
Outlook for Oncosence
Very few public details. Unknown timeline for IND-enabling / clinical trials.
Unknown target indication -- perhaps liver cancer?
Risk
Combination drug therapy means increased chance for off-target effects and drug-drug interactions.
Reward
Screening platform to generate more potential leads and candidates. Can target specific cancers
Targeting cancers with a high unmet need.
Senisca
Funding: unknown
Inception: 2020
Modality: Oligonucleotides, small molecule drugs
Pipeline:
RNA oligonucleotide // Idiopathic Pulmonary Fibrosis // Preclinical
RNA oligonucleotide // Osteoarthritis, dry AMD // Preclinical
Small molecule drug // Skin ageing // Preclinical
Lab Lineage: Lorna Harries
Founders: Lorna Harries
Notable investors: ?
General Notes
Senisca is developing oligonucleotide senomorphic therapies that reverse cellular senescence by modulating RNA splicing factors that become dysregulated with age.
Based on the microRNA-senescence research of founder Lorna Harries, a professor of Molecular Genetics at University of Exeter Medical School.
Targeting senescent-cell related diseases. Lead indications include IPF, osteoarthritis
Also developing a senomorphic pipeline for skin cosmetics.
Origins & Funding
Lorna Harries, CSO and Founder: Professor of Molecular Genetics at University of Exeter Medical School. Main area of research: RNA Splicing factors and aging (Harries et al. 2011).
Recipient of Innovate UK grant. Presently seeking funding for a 950k GBP seed round, whichrepresents a 9-month cash runway.
Company being incubated at University of Exeter (access to lab etc)
Pipeline Details
Developing oligonucleotides (short strands of RNA) to silence specific negative regulators of RNA splicing factors thereby rejuvenating senescent cells.
RNA splicing factors determine how a pre-mRNA sequence will be cut into mRNA, thus allowing a single gene to express more than one mRNA sequence (alternative splicing).
Certain RNA splicing factors levels decrease with age and are linked to cellular senescence (Lee et al. 2016).
Inhibition or targeted knockdown of genes (ERK, AKT, FOXO1, ETV6) that negatively regulate RNA splicing factors reduced senescent cell load and partially reversed multiple cellular senescence phenotypes (SASP, etc) in human fibroblasts in vitro (Latorre et al. 2018)
Lead indication is Idiopathic Pulmonary Fibrosis (IPF), which is linked to senescent cells (Schafer et al. 2017).
IPF is a $2 billion global market with no cure. Current treatments only slow the progression of the disease.
Other planned indications include osteoarthritis, Dry Age-related Macular Degeneration (AMD), and skin ageing.
Oligonucleotide therapies include antisense oligonucleotides (ASO), short interfering RNA (siRNA), and microRNA (miRNA). It’s not clear which sub-modality Senisca is developing.
Proposed RNA delivery vehicles: Lipid vehicles, bare oligonucleotides, nanoparticles.
Outlook for Senisca
Still in early development finding lead targets. Not tested in vivo yet. Candidate selection expected by 2023 EOY. Currently has 4 upstream RNA splicing targets and will validate 20 downstream targets.
Raising a 950k GBP seed round funding, which will give the company a 9-month cash runway.
Currently negotiating deals with a large cosmetics company to license out the skin ageing pipeline.
Risk
Oligonucleotide delivery is a challenge for RNA therapeutics, though there has been much progress in this space (10+ FDA approvals, see Ionis Pharmaceuticals)
Limited cash runway currently. Still very early in development -- no in vivo demonstrations.
Possibility of cancer risk when reversing senescence in some cells?
Reward
Unique senomorphic approach that rejuvenates senescent cells versus merely preventing cells from becoming senescent or killing them. No other competitors in the RNA splicing senolytics niche.
Other companies
There are a number of other companies that I have not included in the above analysis -- mostly because there was not enough public information about them or they were not developing a therapeutic. These include:
OneSkin: Sells a skin cream containing a proprietary senolytic peptide, OS-1. Preclinical and ex vivo studies indicate OS-1 reduces senescent cell burden. But because the cream is marketed as a cosmetic it does not make any therapeutic claims nor has it undergone clinical trials.
Geras Bio: Scott Lowe’s (Memorial Sloan Kettering) startup based on senolytic CAR-T therapy.
NRTK Biosciences: Developing a more potent form of fisetin, a plant flavonoid found in strawberries and other fruits and vegetables. Fisetin is believed to have some senolytic properties.
Eternans: Founded by Osman Ugur Sezerman, a professor at the University of Istanbul. Eternas is developing a peptide that targets FOXO4 instead of the FOXO4-p53 interaction.
1E Therapeutics: Developing antisense RNA senolytics. Couldn’t find much public information about this company other than what was in Elie Dolgin’s Nature article.
Insilico Medicine x Taisho Pharmaceuticals: Insilico Medicine, Alex Zhavoronkov’s AI drug discovery startup, announced a partnership with Taisho Pharmaceuticals to find senolytic drugs.
Recursion Pharmaceuticals: An exciting AI drug discovery / digital biology startup that received an NIH grant to find senolytic compounds and biomarkers. No published results on this yet.
Newomics: Developing a microfluidics assay that can detect and count senescent cells in a blood sample based on cell size.
Velabs: Developing antibody senolytic therapeutics. No other details.
TAmiRNA: Developing a technology to detect senescence-related microRNAs in blood.
Some Observations, Trends, and Predictions
Unity Biotechnology way ahead of the pack: No other company is in the clinic (though Mayo Clinic has a Dasatinib + Quercetin Phase 2 trial). Unity has already completed a Phase 2 (UBX0101, failed) and they are currently conducting a new Phase 1 (UBX1325). But first is not always best — Unity’s approach is somewhat crude compared to the targeted approaches of newer senolytics companies. But perhaps it will be enough.
More senolytic trials are coming: Unity won’t be alone in the clinical stage for long. I believe that FoxBio, Senolytic Therapeutics, Numeric Biotech, and Rubedo Life Science will be in the clinic within 12 - 24 months.
1st Gen vs 2nd Gen: Dasatinib, Navitoclax, UBX0101 were 1st generation senolytics developed by hypothesis: That existing drugs that targeted anti-apoptotic pathways might also clear senescent cells. Unfortunately, many of these 1st gen drugs have off-target effects that kill non-senescent cells, too.
Now it appears the majority of the industry is focusing on 2nd generation therapies through high-throughput screens to improve selectivity. Novel targeted modalities (peptides, mAbs, gene therapy, immunotherapy, RNA) and delivery (nanoparticles and conjugate prodrugs) are being developed as well.More public companies coming?: Unity Biotechnology is the only senolytics company to go public so far. However, Juvenescence is planning to go public within ~6 months, at which point you will be able to own a slice of FoxBio. The current market is amenable to IPOs so I wouldn’t be surprised if another senolytics company went public just before their Phase 1 trials -- perhaps in the next two to three years, should markets hold up.
Senomorphics vs Senolytics: The majority of companies are focussing on killing senescent cells with senolytics. However, four companies have senomorphic pipelines (Senolytic Therapeutics, Senisca, Atropos Therapeutics, Dorian Therapeutics). I’m excited to see which approach will prove most effective, though I am less sanguine on the clinical prospects of approaches that merely slow senescence.
Diversity in indications: The number of different applications for senolytics (and senomorphics) is quite wide: Cancer, age-related diseases, non-age-related diseases -- almost anything related to damaged cells. Most common indications are cancer and fibrotic diseases (especially in the kidney and lung).
Pan-senolytic? The promise of what I like to call the “Grand Unified Theory of Longevity Medicine” is that a therapeutic that works by reversing aging would be applicable to many age-related diseases.
Unfortunately, senescent cells have diverse phenotypes. Some drugs work better on certain senescent cell types vs others. And there is no known universal biomarker for senescence. So while the target (senescent cells) is universal it might be difficult to find a single pan-senolytic that works on all types of senescent cells and their related diseases.But perhaps we don’t need a pan-senolytic. Maybe a poly-senolytic or a combination of different senolytics is sufficient to treat a large swath of age-related diseases.
Would a 2nd Unity Biotechnology failure have an impact on the future of senolytics? Yes and no. Unity is just one of many companies developing senolytics -- and their current clinical trials are only testing the earliest senolytic strategy (1st gen). There are many more senolytics companies aiming for clinical trials in the next two years -- and some will be testing 2nd generation targeted therapies.
If UBX1325 were to fail in Phase 2 the immediate effect would be a decrease in unsophisticated capital in the senolytics space -- from those who don’t understand the other promising targets and modalities in development. This might be a notable amount of money in the short term but long term I am optimistic about senolytics. All it takes is one success for the floodgates to open.
Miscellaneous Notes
Local vs Systemic delivery: Unity Biotechnology is taking the local injection route to deliver their senolytic therapies. They have no choice, considering the toxicity and modest selectivity of first gen senolytics. But what about the next batch of clinical companies? My guess is most will still go local delivery unless they get a breakthrough in highly targeted delivery (prodrugs, nanoparticles, etc)
Healthspan vs Lifespan: I was surprised by the paucity of lifespan studies on naturally-aged mice treated with senolytic drugs. Only Dasatinib seems to have a lifespan result: Xu et a (2018) did a study with Dasatinib + Quercetin that showed old mice had 36% higher post-treatment lifespan compared to controls. Peter de Keizer also seems to have some unpublished results that showed FOXO4-DRI could increase the lifespan of mice by 13%.
There are, however, many senolytics studies demonstrating aging phenotype reversal, improved cognitive function, and physical function. (i.e healthspan), which are more relevant for clinical trials.
Senolytics: Use first, confirm hypothesis later. Senolytics might have tremendous therapeutic benefit. But because we have no easy way of detecting senescent cells in most tissues of patients many of these clinical trials won’t be able to confirm clearance of senescent cells. Unity’s failed UBX0101 did not answer the senescent cell hypothesis (in humans) and the upcoming tranche of trials will not either. However, if we get a string of successful Phase 2 trials we might reasonably infer the senescent cell hypothesis is correct.
Early development takes time. A lot of the smaller companies are very early in development. At this stage they are more like science experiments than drug companies. Don’t expect much (public) guidance on timelines. Taking an initial idea from the lab to the clinic can take 5+ years.
Valley of Funding Death: Most companies are at the seed stage. Anecdotally, there are many investors willing to write million dollar seed stage checks for longevity startups. But if these companies want to get to Phase 1/2 trials they will need to raise much more money (tens of millions).
Steelmanning senolytics. How might they fail? It’s easy to get caught up in the promise of senolytic therapies, especially seeing how well they work in mice. But humans are not mice. My strongest argument against senolytics would be that we only really have mice studies as evidence for rejuvenation. I guess that is par for the course for most drugs. I couldn’t find any senolytic rejuventaion demonstrations on other mammals in the literature. It might turn out that mice just respond differently to senescent cell ablation than humans — though this seems unlikly and ad hoc. Perhaps the super long telomeres of mice play a role in their observed senolytic-related rejuvenation. Just speculation — I have no idea. My worry is that the upcoming clinical trials for senolytics fail but we don’t know whether it was because the therapy didn’t clear the target senescent cells or it was because clearing senescent cells doesn’t have therapeutic benefit.
Database (in progress)
Senolytics companies database: